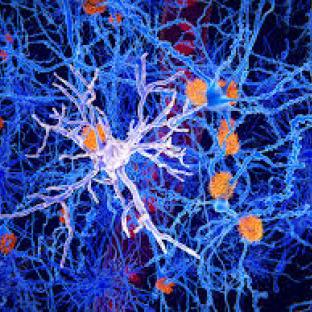
On 2 November, Neuraly, a clinical stage biopharmaceutical company translating scientific discoveries into revolutionary new drugs for neurodegenerative disorders such as Alzheimer’s disease (AD) and Parkinson’s disease, announced it has received clearance from the US Food and Drug Administration (FDA) to initiate a Phase IIb clinical trial of NLY01 in AD. NLY01 works by activating a protein called glucagon-like peptide 1 receptor (GLP-1R). This protein is highly expressed on microglia, which are immune cells that reside in the brain. The Phase IIb trial is multicenter, randomised, double-blinded and placebo-controlled study assessing the safety, tolerability and efficacy of NLY01 in people with mild cognitive impairment due to AD. The company expects to recruit more than 500 research participants in the US, Canada and Europe. Topline results are expected by Q4 2023.
Neuraly receives FDA clearance to initiate Phase II trial of NLY01 in AD
02/11/2020